Precision Medical Care Newsletter March 2026
Doctor Agarwal and Johnson of Precision Medical Care attend the December 2025 meetings at the Buck Institute of Aging And Research, Longevity Clinics Roundtable and Longevity Summit conferences
From the desk of Dr Catherine
2026 is flying by and has been very productive, the practice volume has been growing steadily and we are most grateful for every referral that allows us to expand access to the benefits of Precision Medicine. Thank you!
Dr. Agarwal and I wrapped up 2025 with our colleagues at the Buck Institute Research on Aging. We attended both the Longevity Summit and Roundtable of Clinics. Here is a precision link to the Buck Institute conference discussions, there are so many exciting advances coming in the longevity medicine community, especially as AI applications are accelerating access and scale: https://drive.google.com/drive/mobile/folders/1jmsKrhwXYZIrhjLQYyhfylslMd2EdInQ?usp=sharing
It is with great pride that I can share this news: Precision Medical Care’s research has been submitted for publication to the medical journal Frontiers in Endocrinology. “Effects of GLP-1 Receptor Agonist Therapy on Body Weight and Body Composition in Adults with Overweight and Obesity.” Our research is among the first to be published reviewing results from private practice that out-performed all published clinical trials, also the first paper to present data from bio-impedance analysis (vs DEXA) for body composition testing.
The paper retrospectively reviews 34 adults (average age 53) who were being treated for excess weight/overweight/obesity at a medical clinic. Patients took tirzepatide once weekly for an average of about 1.5 years while also receiving precision medical care: medical prescriptions, diet, lifestyle, supplement and exercise guidance under the care of Dr Johnson, an obesity medicine certified physician.
The key finding: Most of the weight lost was fat, not muscle.
• On average, participants lost about 28% of their body weight
• About 86% of the weight lost came from fat
• Only about 14% came from lean tissue (muscle and water)
• Body fluid balance stayed healthy throughout treatment
Latest Updates in Cardiovascular Risk Assessment and Weight Management Therapies: AHA PREVENT Calculator: A New Approach to Heart Health
The American Heart Association recently released updated guidelines featuring the PREVENT calculator. Notably, a longevity approach is showing up in the guidelines: what is the estimated ten year risk AND what is 30 year risk of heart disease. We want to be around for a good time and a long time!
As we’ve mentioned in previous newsletters, updated guidelines are including biomarkers related to blood sugar regulation, kidney function and zip code! These factors provide more precise risk estimates by including kidney function and metabolic factors.
Risk thresholds have been adjusted: borderline risk is now 3-5%, intermediate risk is 5-10%, and high risk is 10% or above. You can calculate your risk and correlate with LDL targets based on these updated guidelines.
https://professional.heart.org/en/guidelines-and-statements/prevent-calculator.
Oral Wegovy (Semaglutide): GLP1 agonist in a weight loss pill
Great news for those who prefer not to use injections! The FDA has approved an oral tablet form of Wegovy (semaglutide) at a 25 mg daily dose for weight management.
Clinical trial results showed:
Average weight loss of 15.1% at 64 weeks with the oral tablet
92% of participants lost at least 5% of their body weight
75% lost at least 10% of their body weight
The medication also reduces cardiovascular risk in people with heart disease and obesity
The oral tablet is taken once daily and offers an alternative for those who prefer pills over weekly injections. Both forms are effective when combined with reduced-calorie diet and increased physical activity.
Notably, lower doses of 1.5 mg Wegovy are only $149 for 30 day supply and this dose has similar bioavailability to Rybelsus at 3 mg dose (another oral semaglutide which has been FDA approved for many years for type 2 DM ). The cash price for Rybelsus is 10x as high as oral Wegovy.
Semaglutide has been studied extensively and is very safe in humans up to a dose of 7.2 mg injectable weekly. The higher dose injectable Semaglutide is now FDA approved as of March 19th! At the highest dose, there is a bit more weight loss too- getting close to the efficacy of Tirzepatide.
Zepbound (Tirzepatide) Updates
Zepbound continues to show impressive results for weight management. Recent data confirms:
Average weight loss of 20-24% of total body weight at 72 weeks depending on dose
Up to 93% of people lost at least 10% of their body weight on the highest dose
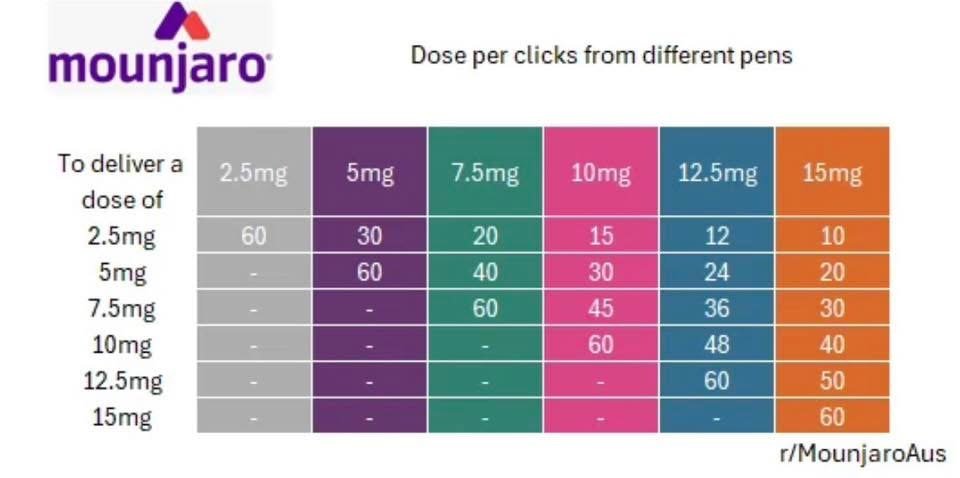
LillyDirect released the Zepbound KiwikPen. The KwikPen is one pen that holds all four weekly Zepbound doses for the month. You can learn more about the KwikPen HERE.
For reference, the KwikPen has been available in other countries for Mounjaro precision dosing. One pen can hold several doses and the click to dose reference ranges can be helpful when making dose adjustments. You will add a fresh/sterile needle tip to the Qwikpen with each injection, similar to the Ozempic multi dose pen.
Vybrique: breaking news that no one seems to know about…
a faster acting formulation of Viagra?
Vybrique (sildenafil) is an FDA-approved, fast-acting oral film for treating erectile dysfunction (ED) in men, developed by IBSA USA. It dissolves on the tongue without water, providing a discreet,, on-the-go option. It is available in 25, 50, 75, and 100 mg doses. Only available for cash pricing $8 a dose via specialty pharmacy.
Precision Peptides
Social media has been buzzing about various peptides and supplements. We have had Retatrutide on our radar for years as it’s been studied as the triple G peptide: agonist of GLP1, GIP and Glucagon. Glucagon is the “third G” to this tri-peptide and adds potency to the glucose lowering and weight loss lowering effects of Tirzepatide.
Retatrutide: This is an investigational triple-hormone receptor agonist currently in clinical trials (not yet FDA-approved). Early research shows promising weight loss results of up to 24% at 48 weeks, but it's only available through clinical trials at this time. It's not the same as the FDA-approved medications Wegovy or Zepbound. We expect this “triple G” peptide to be on the market later this year. As you might expect, we will have samples of the legal Retatrutide at the office as soon as it’s available and we will prescribe it for you based on your candidacy for the medication. If you have heard of others “taking Reta,” please ask them to check their vial (likely sourced via a gym bro or a TikTok link) as it is likely marked as “for research purposes only, not for human use.”
NAD+ Supplements: NAD+ (nicotinamide adenine dinucleotide) is a molecule that declines with age. Oral supplements like nicotinamide riboside (NR) and nicotinamide mononucleotide (NMN) are being studied for anti-aging effects. Early research suggests they may help with blood pressure, insulin sensitivity, and inflammation, but large-scale clinical trials are still needed. These supplements appear safe but their long-term benefits in humans remain uncertain. Thorne’s Resveracel is likely a reputable choice and available at our offices if you’d like to trial NAD “boosting” supplements. The injectable NAD formulations are available for prescription, also.
Glutathione: This is your body's most abundant antioxidant. Some studies show oral glutathione supplements can increase blood levels and may improve insulin sensitivity and reduce oxidative stress markers. The precursor to glutathione is NAC, N acetyl cysteine. This is a supplement we have at the office and it is also included in a formulation “Liver Protect.” Injectable glutathione is available by prescription, too.
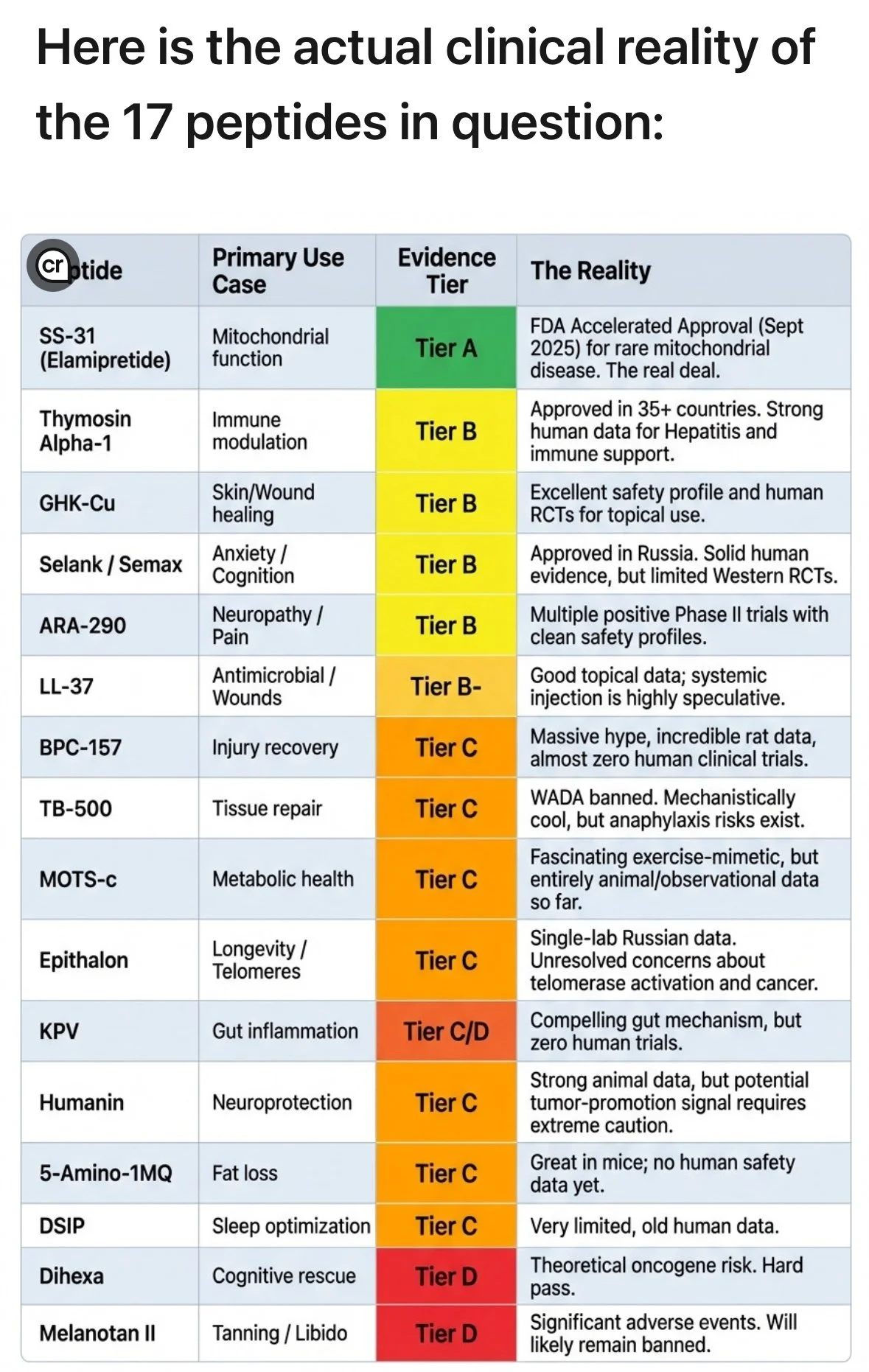
A visual review of the FDA’s list of peptides under clinical review and study based on tiers of safety. Here is a breakdown of the safety tiers for investigational peptides:
Tier A. FDA-Approved Peptides (Highest Safety Tier) These are approved for specific human conditions, manufactured under strict GMP (Good Manufacturing Practices) guidelines, and have undergone rigorous clinical trials. Examples: Semaglutide, Tirzepatide (GLP-1s), Tesamorelin, Sermorelin. Status: Highest safety profile due to quality control and known human data.
Tier B. Compoundable Peptides (Under FDA Discretion) These peptides are not specifically FDA-approved but are often allowed to be produced by licensed 503A or 503B compounding pharmacies under FDA guidance. Examples: Thymosin Alpha-1, BPC-157 (previously), certain GHRHs. Status: Moderate risk. While produced by licensed pharmacies, they lack the full FDA approval process for efficacy and long-term safety. Regulatory Shift: Many, such as BPC-157 and Thymosin Beta-4, have been moved to Category 2 (restricted) by the FDA because they may present significant safety risks or lack enough data.
Tier C. "Research Only" / Non-Approved Peptides (High Risk) These are bought from online vendors and labeled "not for human consumption." They are frequently manufactured in unregulated facilities without quality control, resulting in high potential for contamination or incorrect dosage. Examples: BPC-157 (purchased online), TB-500, Epitalon, Dihexa. Status: High risk. These substances lack rigorous sterility testing, purity checks, or controlled human trials.
Tier D. Banned/Category 2 Peptides These are materials that the FDA has specifically identified as presenting significant safety risks, prohibiting them from being used in compounding.
Unregulated Peptides: Why they are “Research Only” for humans:
Key Safety Considerations for “Black Market” peptides that are minimized on social media:
• Immunogenicity: An adverse immune response can occur, triggered by the peptide itself or impurities, potentially causing serious side effects.
• Theoretical Risks: Many non-approved peptides lack long-term data on potential risks like aberrant tissue growth or effects on dormant malignancies.
• Purity Issues: Many "research-only" peptides are not tested for potency or contamination, with online products often failing quality checks.
• Administration: Self-administration of research peptides increases the risk of injection site reactions and dosing errors.
In the News: Galleri/Grail test
A recent study was published related to the “liquid biopsy” Galleri test which highlighted some limitations of the test. The trial showed several important and encouraging results:
More than 4 times the number of cancers were detected when adding the Galleri® test to standard-of-care screening.
More cancers were found earlier, reducing stage IV diagnoses vs. standard-of-care screening.
Strong test performance, consistent with prior clinical studies
No serious safety events arose
What was the NHS-Galleri trial?
Here are important facts about the NHS-Galleri trial 1:
Trial design: The first and only randomized controlled trial of a multi-cancer early detection test. Randomized controlled trials are the gold standard of evidence in the healthcare industry.
Collaboration: The trial was conducted in partnership with the National Health Service (NHS) in England, the publicly funded, government-run healthcare system.
Scope: A very large research trial that enrolled 142,000 people ages 50 to 77.
Purpose: To determine whether adding the Galleri multi-cancer early detection test to standard-of-care cancer screening in England could help detect more cancers earlier.
Timeframe: Trial participants took a Galleri test annually for 3 years and were followed for 1 year after their last blood draw. You may have seen the headlines and media reports. In addition to the results noted above, the NHS-Galleri trial missed its primary endpoint.
Here’s what that means:
Designed as a screening trial in collaboration with the NHS in 2020, the main goal was to understand if the Galleri test could reduce the incidence of late-stage cancers. A combined stage III-IV reduction following 3 years of annual screening was chosen as the primary endpoint at the time because it most closely aligned with the NHS long-term plan, which aimed to diagnose more cancers at stage I-II.
That goal was not met. While there was a favorable trend toward a reduction in stage III and IV diagnoses at the end of the trial, there was no statistically significant reduction in the combined rate of stage III and IV cancers after screening annually for 3 years.
Stage IV reduction was selected as a key secondary endpoint because the value and importance of a stage IV reduction alone is well-recognized.
The trial did find that stage IV cancer diagnoses decreased with each year of screening, with a greater than 20% reduction in the second and third years of screening with the Galleri test.
Here are some visual highlights of the study: https://engage.grail.com/viewer/5bade164c61509102a06735a688e98bb#1
Podcasts & Events
Dr. Catherine spoke with Dr. Shauna Watts recently on her podcast about women’s health and hormones. Listen here.
Dr. Catherine will be speaking at an upcoming Women’s Health event in Miami on April 25th hosted by Dr. Jila and the Miami menopause collective. We’d love to have you join if you’re interested.
Precision Lab interpretation and trending over time is our specialty.